Our current research focuses on bioengineering and therapeutic targeting of the bone marrow microenvironment that implicate clinical importance as a major reservoir of adult stem cells that are in constant production of mature blood cells, a key regulator of body homeostasis via continuous crosstalk with distant organs, and in instances of cancerous lesion, a potent instigator of metastatic spread. With core expertise in biomaterials, microfabrication, cellular engineering, and biomedical imaging, we study underlying roles of bone marrow extracellular matrices and stromal cells in forming and sustaining highly regenerative microenvironments as well as their transformation in pathological and aging processes. The science and technology under the study are expected to contribute to broad areas of research including regenerative medicine, cell transplantation, stem cell niche targeting therapeutics, tumor metastasis, immunotherapy and aging. Our research will gradually expand to create other lymphoid tissue analogues (e.g. lymph nodes, thymus, spleen) while pursuing extensive collaboration to drive adoption of these models by basic, clinical and industrial researchers in stem cell and cancer biology.
Bone Marrow in Health and Disease

HSC: Hematopoietic Stem Cells
BMDC: Bone Marrow Derived Cells
CTC: Circulating Tumor Cells
Microphysiological Bone Marrow Organoids
Creating functional bone marrow organoids outside of the body represents great opportunities in many fields of modern medicine while remaining as a challenging task due to its tissue complexity. From structural and functional aspects, bone marrow can be divided into three distinct microenvironments i.e. vascular, interstitial and bony surface. Each compartment has an intrinsically different cellular and extracellular composition that ultimately results in different mechanical properties of the tissue while functioning as a single organ. In order to recreate such complex bone marrow microenvironments in a controlled and manipulatable manner, we aim to develop multi-compartment tissue models via integrating separately constructed and optimized individual modules in a single platform. Established microphysiological tissue models are expected to greatly enhance the predictive power of preclinical testings. Ultimately we aim to create personalized preclinical tissue models by leveraging recent advances in induced pluriopotent stem cells (iPS) research, which can closely represent patient specific tissue microenvironments. Such personalized in vitro tissue surrogates would rapidly and precisely guide the most effective treatment.
Anatomical & Material Aspect of Bone and Bone Marrow

Synthetic & Analytical Marrow Mimicking Microenvironments
Inverted colloidal crystal hydrogel scaffolds

Ectopic Skin Marrow
Despite its clinical importance, bone marrow located inside of bony cavities is limited in study because of its anatomical inaccessibility and tissue complexity. Leveraging tissue engineering principles and immunodeficient mice models, we aim to create humanized marrow mimicking microenvironments beneath the skin. Standardized, analytical and experimentally accessible ectopic skin marrow can facilitate various bone marrow related studies including hematopoeitic stem cell trafficking, extramedullary hematopoiesis, and red-to-yellow marrow conversion.
Implantable microenvironments forming vascularized HSC niches

Capture dynamic cellular process at real-time, single cell resolution

Bone Marrow Metastasis
Tumor cell metastasis is an insidious phenomenon that causes 90% of cancer-associated death while remaining the most poorly understood aspect in carcinogenesis. Bone marrow is one of major metastatic sites for prominent tumor types including prostate and breast tumors. Utilizing manipulatable and transplantable bone marrow tissue models, we aim to determine vital molecular and cellular components that are the linchpin of tumor metastasis, which can be potentially translated into therapeutic targets within the bone marrow niche.
Enabling bioengineered microenvironments for metastasis research
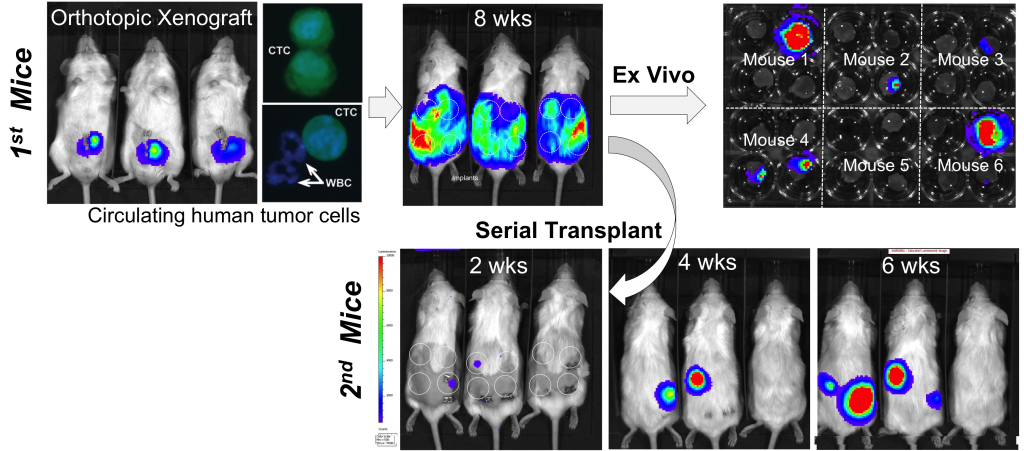
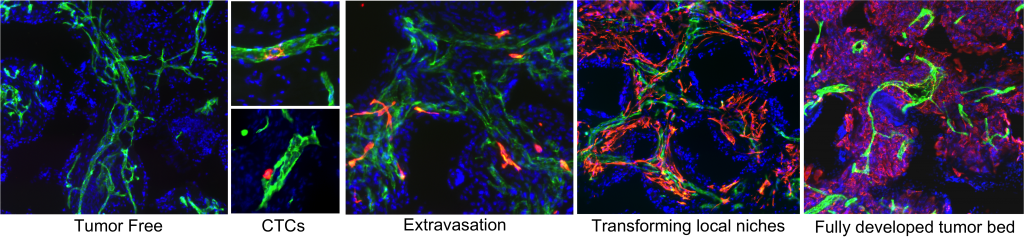
Recapitulating the full spectrum of metastatic cascade
Therapeutic Targeting Bone Marrow Niche
Many conventional stem cell and cancer therapies primarily focus on the target cells of interest in the bone marrow; however, there is now a greater appreciation for targeting the other components in the microenvironments to indirectly modulate stem and cancer cell fate. To realize such “microenvironment-targeting” approach, we aim to develop drug delivery systems that can selectively target specialized bone marrow microenvironments and subsequently release payloads in a controlled manner.
Tissue Engineered In Vitro Drug Testing Platform
Besides bone marrow, we explore bioengineering other important tissues such as hepatic liver spheroids for basic and translational research.
Standardized hepatic spheroid culture model in ICC hydrogel scaffolds
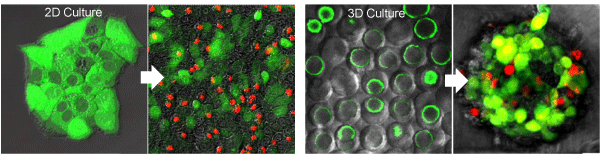
Comparison of nanoparticle toxicity testing in 2D vs. 3D culture
Microfluidic Tissue Models
Integrating soft biomaterials and microfabrication, we also actively pursue to create in vitro microfluidic tissue models. Previously we combined a 3D hydrogel scaffold and PDMS-based fluidic chip for creating microfluidic marrow-mimic that can capture perfused human cancer cells, support growth, and further retrieval and xenograft tumor formation.
SPONSORS


